يمكن للطاقة الحرارية الانتقال بين النظام system والوسط المحيط surroundings من خلال طريقتين هما:
من خلال الشغل Work وهذا يؤدي إلى تغيير في الضغط والحجم ودرجة الحرارة ويقال عن هذا التغيير أنه من تغيير في الحالة الجاهرية Macroscopic.
من خلال التدفق الحراري Heat وهذا يؤدي إلى تغيير في الطاقة الداخلية للنظام ويقال عن هذا التغيير أنه تغيير في الحالة الجوهرية Microscopic.
If a system (gas) in its initial state at Pi, Vi change to Pf, Vf by adding quantities of heat and applying work on the system. If the quantity Q-W is measured for various paths from i to f. we find
Q-W is always constant.
Q-W is called the change in the internal energy of the system ΔU
Q and W depend on the path but Q – W is independent of the path
ΔU = Uf – Ui = Q – W The first law of thermodynamics
for small changes
dU = dQ – dW
Special cases
In isolated system there is no heat flow and work is zero the change in internal energy is zero, i.e. DU=0
If the process is done on a system taken through a cycle, the change in the internal energy is zero, i.e. ΔU=0 and Q = W
Example
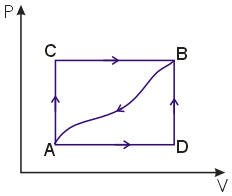 When a system is taken from state A to State B along the path ACB, 80J of heat flow into the system and the system does 30J of work,
When a system is taken from state A to State B along the path ACB, 80J of heat flow into the system and the system does 30J of work,
(a) How much heat flow into the system along the path ADB if the work done is 10J.
(b) The system is returned from the state B to A along the curved path. the work done on the system is 20J
(c) If UA=0, UD=40J, find the heat absorbed in the process AD and DB.
Solution
(a) Along the path ADB
W = 10J
UB – UA = QADB – W
Along the path ACB
UB-UA = QACB – W
= 80 – 30 = 50J
hence
QADB = 50 + 10 = 60J
(b) for the curved path from B to A
W = -20 (شغل بذل على النظام)
Q = (UA – UB) + W
= -50 – 20 = -70J
(The negative sign show that heat is liberated by the system)
(c) UA = 0 & UD = 40J & UB – UA = 50J then UB = 50J
QAD = (UD – UA) + W
In the process ADB, 10J of work is done = work done from A to D is 10J and from D to B is zero (at constant volume)
for AD QAD = 40 + 10 = 50J
for DB QDB = (UB – UD) + W
= (50 – 40) + 0 = 10J
Example
A thermodynamic process is shown in Figure. In process ab, 600J of heat are added, and in process bd 200J of heat are added. Find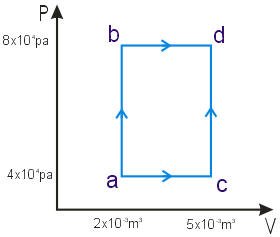
- the internal energy change in process ab
- the internal energy change in process abd
- the total heat added in process acd
Solution
(a) in ab W = 0 and ΔU = Q = 600J
(b) in bd pressure is constant
W = P (V2 – V1) = 8×104 pa (5×10-3 – 2×10-3) = 240J
Wabd = 240 + 0
Qabd = 800J
ΔU = 800 – 240 = 560J
(c) in acd ΔU = 560J as well
W = 3×104 pa (5×10-3 – 2×10-3) = 90J
Q = ΔU + W = 560 + 90 = 650J
Problem (1)
what is the final temperature of two system a and b are in thermal contact and both a and b are insulated, prove that
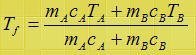
Hint QA + QB = 0
Problem (2)
The volume of an ideal gas is 4 litter, the pressure is 2 atm and the temperature is 300K. The gas first expands at constant pressure to twice its original volume, it is then compressed isothermally to its original volume and finally cooled at constant volume to its original pressure.
(a) show the process on a pv diagram
(b) Compute the temperature during the isothermal compression
(c) Compute the work done by the gas during the expansion
(d) Compute the maximum pressure attained in the process.



